Have u ever noticed that winters are not so cold now-a-days!
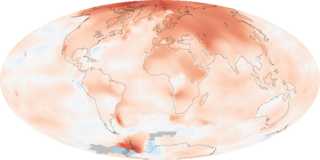
The main cause of this is Global warming!-Global warming is the rise of the temperature on the surface of Earth. AND what is causing this rise in temperature? WE HUMANS OF-COURSE!
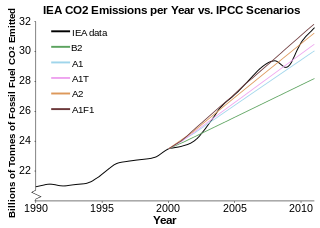
The cause of the warming is the GREENHOUSE EFFECT- gases of organic chemicals(Methane, Carbon dioxide etc) when released in the atmosphere absorb a lot of heat and also stop the heat of Earth from escaping it that make Earth hotter. emission of these gasses are increasing day by day.
The main cause of this is Global warming!-Global warming is the rise of the temperature on the surface of Earth. AND what is causing this rise in temperature? WE HUMANS OF-COURSE!
The cause of the warming is the GREENHOUSE EFFECT- gases of organic chemicals(Methane, Carbon dioxide etc) when released in the atmosphere absorb a lot of heat and also stop the heat of Earth from escaping it that make Earth hotter. emission of these gasses are increasing day by day.
only ways to decrease is the control of these emission and growing forest.
if we don't take action, we are only to suffer!!